STAT3
STAT3 in Inflammatory Disease
The interleukin cytokines IL-23 and IL-6 signal through STAT3 and promote the generation and function of pathogenic T helper type-17 (Th17) cells, a type of immune cell that is pro-inflammatory. They are considered pivotal players in certain inflammatory diseases, including rheumatoid arthritis, psoriasis, inflammatory bowel disease and others. Monoclonal antibody therapies that target individual cytokine mechanisms have been approved as treatments; however, these medicines require injection, are often restricted to subsets of patients, and are slow to clear from the body if treatment discontinuation is required.
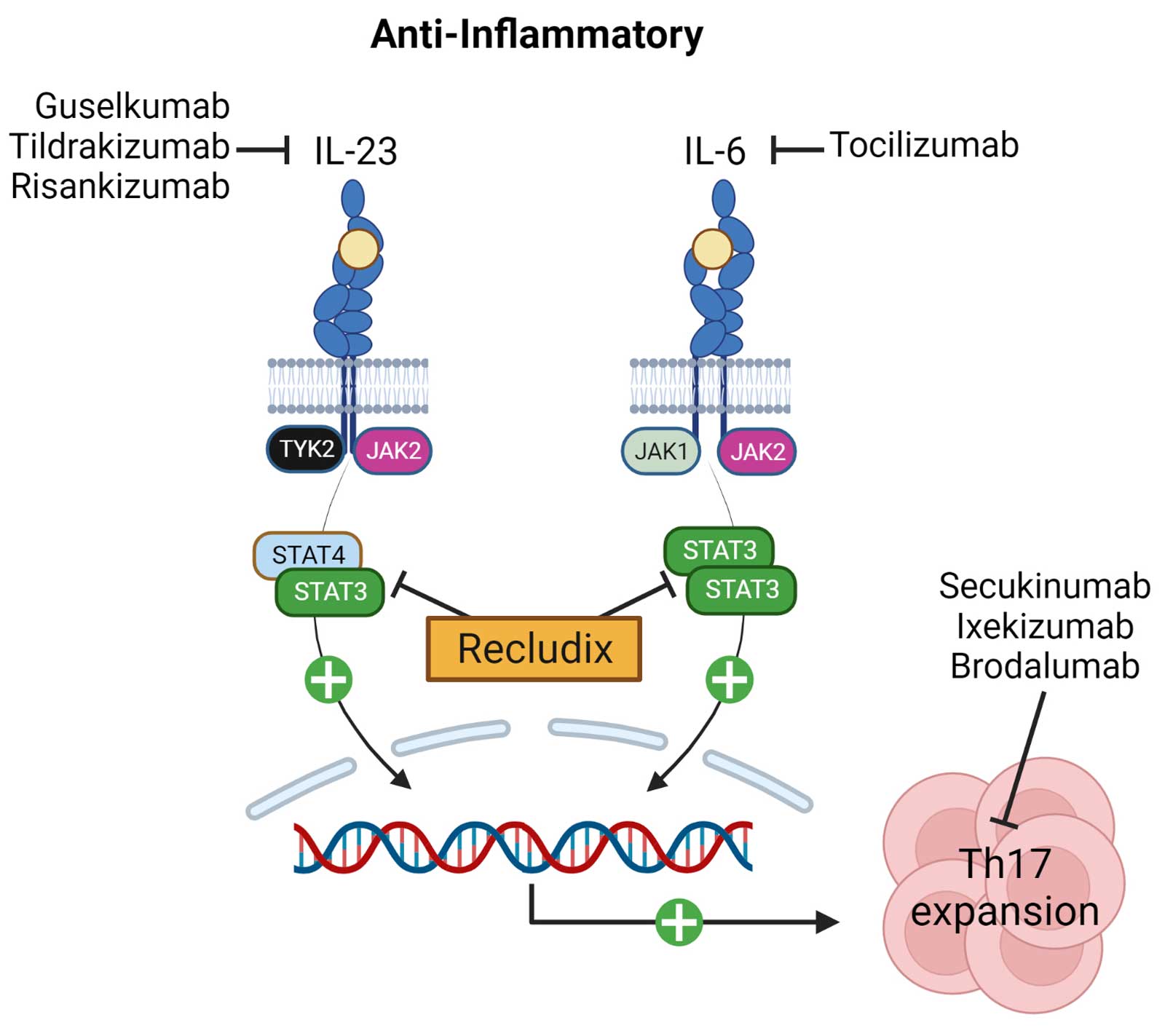
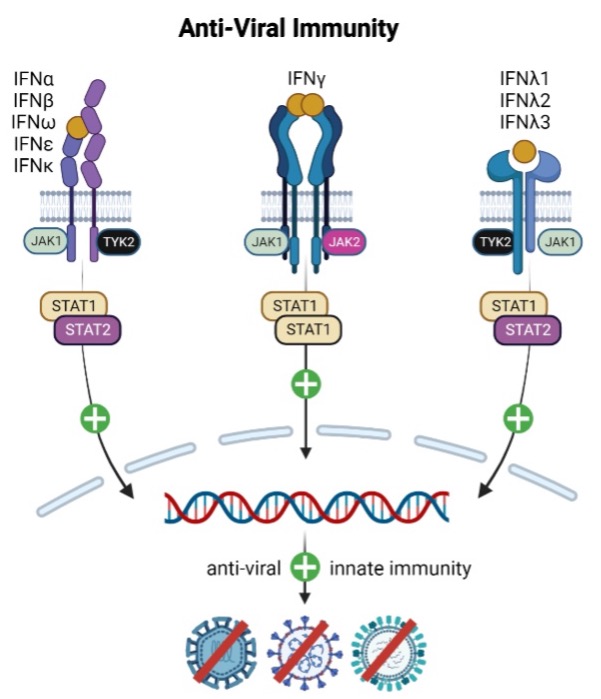
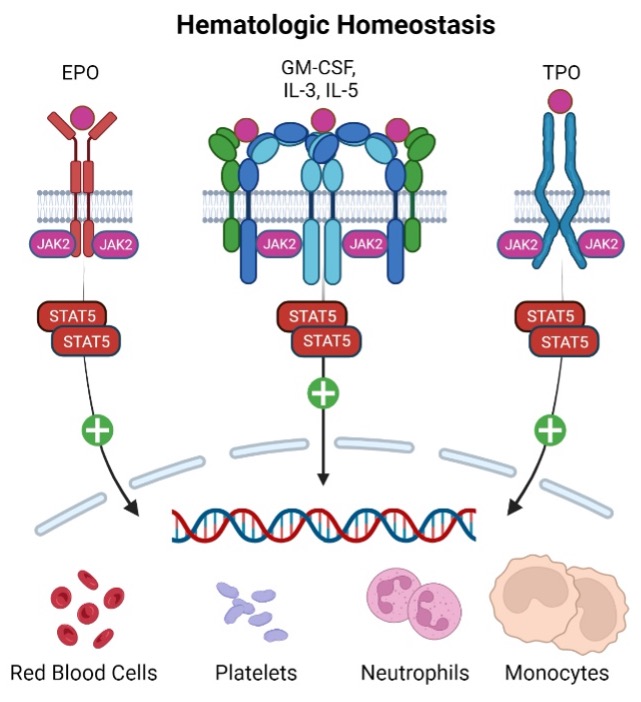
Oral, reversible therapies inhibiting the Janus Kinase (JAK) family, including TYK2, have demonstrated clinical efficacy in Th17-driven diseases. However, JAK family inhibition can be associated with changes in immune surveillance and hematopoiesis, leading to dose limiting safety concerns. Recludix’s therapeutic approach targeting STAT3 is expected to be safer in this regard, as cytokines critical to anti-viral immunity and hematologic homeostasis do not utilize STAT3 signaling.
Targeted STAT3 inhibition with an oral, reversible, small molecule drug represents an attractive alternative to biologics and to JAK family kinase inhibitors, including TYK2 inhibitors, to inhibit Th17 cell activity and treat Th17-driven inflammatory and autoimmune diseases. Importantly, STAT3 inhibition offers potential efficacy beyond Th17 / IL-17 driven diseases in clinical indications where blocking the IL-6 pathway is clinically validated.
STAT3 in Cancer
More than 70% of human cancers exhibit abnormally elevated STAT3 activity and levels of phosphorylated STAT3 have been shown to correlate with poor clinical outcomes in several cancers.
Recurring, activating hotspot mutations are found in STAT3 in multiple leukemias and lymphomas. These include large granular lymphocytic leukemia, about 50% of which harbor STAT3 mutations, with STAT3 activated in virtually all patients regardless of STAT3 mutation status, and diffuse large B-cell lymphoma, with a STAT3 mutation frequency of approximately 5%.
STAT3 mutations in these diseases are largely mutually exclusive with other known oncogenic mutations, strongly suggesting that STAT3 mutations are oncogenic drivers. Tumors with these mutations therefore are likely to be sensitive to STAT3 inhibition.
STAT3 also plays a pivotal role in tumor-infiltrating immune cells that comprise the tumor microenvironment (TME). Hyperactivation of STAT3 in the TME results in immunosuppression by inhibiting both innate and adaptive immune responses, including through the accumulation and enrichment of immunosuppressive Treg cells and the polarization of M2-macrophages, which contribute to immune evasion. Additionally, STAT3 activation is a driver for increased expression of immune checkpoint molecules (such as PD-L1, PD-L2 and CTLA-4) in these cells. Inhibition of STAT3 in this context has the potential to improve the anti-cancer immune response.
